PREMOM II – A remote monitoring follow-up program for women with a high risk for the development of gestational hypertensive disorders
*Project funded by FWO-TBM fund.
Study goal: The goal of this project is to thoroughly evaluate the added value of a telemonitoring (TM) program for women at risk for gestational hypertensive disorders (GHD), by investigating its impact on prenatal follow-up, health outcomes for mother and child, costs and satisfaction, and by specifically investigating what are the major contributors to this added value.
Study design: The study is a multicenter, prospective, interventional randomized controlled trial in which 6107 pregnant women at risk for GHD will be included. Eligible pregnant women at risk for GHD will be recruited after routine GDH screening and randomly divided in one of the following three groups, in each participating hospital:
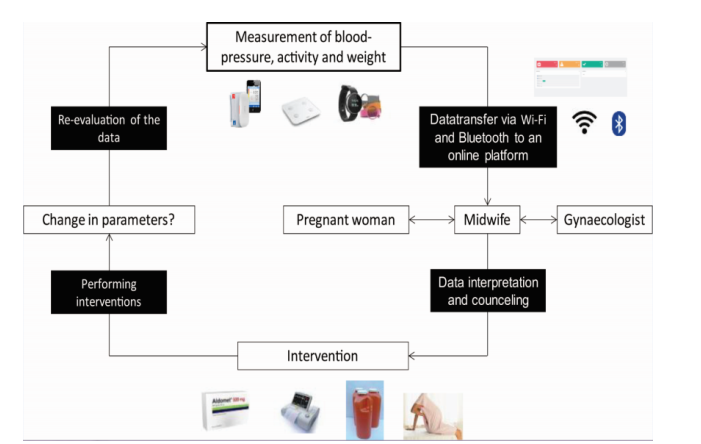
- TM (telemonitoring) group. Pregnant women participating in this group will receive a connected blood pressure monitor and activity tracker. The patient has to measure her blood pressure twice a day (in the morning and in the evening) and fill in her weight once a week in the App. These data are automatically transmitted via Bluetooth and Wi-Fi to a TM-midwife in the hospital who can follow-up those data in a digital dashboard. When abnormal events occur, the referring gynaecologist and the TM gynaecologist will be contacted by the midwife. The TM gynaecologist can give advice concerning the interventions which can be performed but the referring gynaecologist makes the final decision (and will make the prescriptions when necessary). The midwife will use multi-model contacts (i.e. in-person or telephone counseling contacts with the women, face-to-face visits with the pregnant women, etc.) to deliver the intervention to control the blood pressure.
- Patient self-monitoring group. Pregnant women participating in this group will also receive a connected blood pressure monitor. They are asked to collect their blood pressure twice a day and put in their weight on the app once a week. As with the TM group, those data will be transmitted by Wi-Fi and Bluetooth to an online dashboard to store and collect them, but they will not be revised by a healthcare collaborator, nor will the women in this group be motivated to keep on measuring in case their compliance would drop. The women in this group will receive standard prenatal care or will receive interventions when they contact their caregivers about their measurements.
- Conventional care group. Pregnant women in the conventional care group will receive the standard prenatal care, as provided to local sites. They don’t receive any devices.
The following maternal intensive care centers will participate in this project:
- UZ Leuven (Leuven, Belgium; ca 2000 deliveries/year) which reaches ca 9300 deliveries/yr in the wider area, including surrounding hospitals – Prof. Dr. Roland Devlieger.
- UZ Antwerpen (Antwerpen, Belgium; ca 1000 deliveries/year) which reaches ca 1000 deliveries/yr in the wider area, including surrounding hospitals – Prof. Dr. Yves Jacquemyn
- Sint Lucas & Sint Jan Brugge – Oostende (Brugge, Belgium; ca 2000 deliveries/year) which reaches ca 11000 deliveries/yr in the wider area, including surrounding hospitals – Dr. Hilde Logghe
- Ziekenhuis Oost-Limburg (ZOL; Genk, Belgium; ca 2000 deliveries/year) which reaches ca 7600 deliveries/yr in the wider area, including surrounding hospitals – Prof. Dr. Eric De Jonge
Study results: Our goal is to roll out TM in prenatal care for women at risk for GHD in the whole of Flanders. When TM would be applied at all Flemish maternal intensive care centers, almost every women will have access to this prenatal care program which can improve the outcome for her and her baby. The overall goal of this randomized controlled trial is to thoroughly evaluate the added value of a TM follow-up program for pregnant women at risk for GHD. We hypothesize that a prenatal follow-up program complemented with TM will improve the gestational outcomes for both the pregnant women as the neonates, without increasing the costs of healthcare.
With PREMOM II, we indeed address the following triple aim for the prenatal care of high risk GHD pregnancies:
- Improve health (less complications for mother and child because of GHD)
- Improve quality of care (early detection of (exacerbation of) GHD) and more intensive follow-up for those patients that need it.
- Reduce health care costs per capita (lower care consumption – fewer consultations and admissions, fewer costs because of complications for mother and child)
If successful, these results will convince caregivers, hospital management and payers to translate them into clinical practice, and hence implement TM for women at risk for GHD in Flemish healthcare.
Keywords: Gestational hypertensive disorders – remote monitoring – pregnancy outcomes
News
https://www.hbvl.be/cnt/dmf20180919_03762217/helft-minder-zwaar-zieke-prematuurtjes